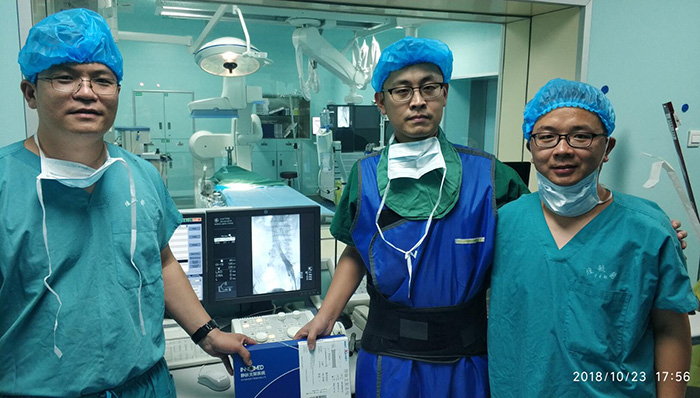
November 2nd, 2018 – Innomed’s new product, Inno-Xmart venous stent system, was used to complete two clinical scientific research operations successfully in Yuhuangding Hospital, Yantai, Shandong Province, P.R. China.
On October 23 and October 28, 2018, the Innomed new product, Inno-Xmart venous stent system, was used successfully to complete 2 clinical scientific research operations in Yuhuangding Hospital, Yantai, Shangdong Province.
The Inno-Xmart venous stent system is mainly used in the iliofemoral vein to treat symptomatic venous obstruction.
Two patients initially diagnosed with pulmonary embolism secondary to deep venous thrombosis in the lower limb were implanted with the Inno-Xmart venous stent system. Postop angiography showed the stent conformed well to the vessel with no residual stenosis and good blood flow inside the vein. Both patients were in good condition after the operation.
Experts say that the Inno-Xmart venous stent from Innomed performs well and is comparable to similar imported products. The operational success once again reflects the Innomed’s R&D concept of “coming from the front line of clinical practice and retuning to the clinic to provide better solutions for patients.”